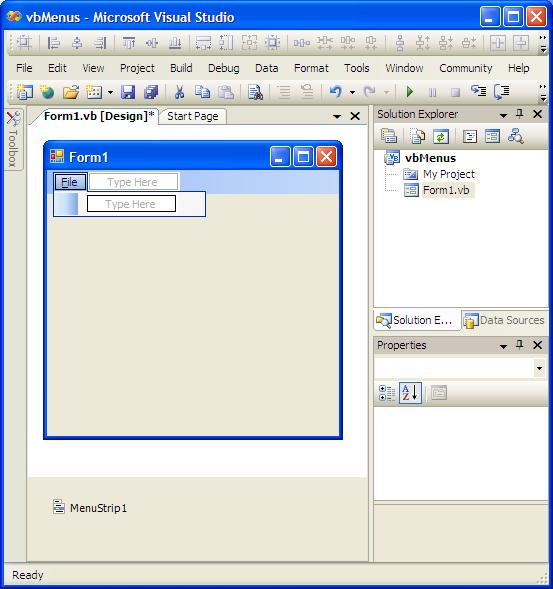
Effects of Silver Nitrate and Silver Nanoparticles on a.
Carbonate ions, CO 3 2-can be detected whether in a solid compound or in solution. An acid, such as dilute hydrochloric acid, is added to the test compound. Carbon dioxide gas bubbles if carbonate.
A reluctant first-order substrate can be forced to ionize by adding some silver nitrate (one of the few soluble silver salts) to the reaction. Silver ion reacts with the halogen to form a silver halide (a highly exothermic reaction), generating the cation of the alkyl group. Give mechanisms for the following silver-promoted rearrangements.
Therefore, this experiment was designed to study the effect of nano silver and silver nitrate on abscission and yield of seed in borage. The study was carried out in a randomized block design with three replications. Four levels of either silver nitrate (0, 100, 200, and 300 ppm) or nano silver (0, 20, 40, and 60 ppm) were sprayed on borage plant at seed growth stage. The results showed that.
If you would like to know more about how silver nitrate behaves, check out our previous blog post where we’ve covered its properties, production, other uses and hazards. Silver Nitrate in History. Records show that silver nitrate as a medical treatment has been used as early as 4000 B.C.E. It was used extensively in places like Ancient Greece.
Lab on Silver Nitrate. Topics: Halogen, Periodic table, Chemical element Pages: 1 (282 words) Published: February 25, 2014. Families of elements—Nonmetals 1. Chemical properties of the halogen family are that the halogens are located in Group VIIA of the periodic table, and are a class of nonmetals. The halogen elements are fluorine, chlorine, bromine, iodine, astatine, and ununseptium.
Silver nitrate is usually prepared by combining silver with nitric acid. Common silver objects used in these reactions include silver bullions and silver foils. The products formed in this reaction include silver nitrate, water, and nitrogen oxides. The byproducts of this chemical reaction depend on the nitric acid concentration that is used. It is important to note that this reaction must be.
Silver nitrate, caustic chemical compound, important as an antiseptic, in the industrial preparation of other silver salts, and as a reagent in analytical chemistry. Its chemical formula is AgNO3. Applied to the skin and mucous membranes, silver nitrate is used either in stick form as lunar caustic.